This is really cool. You know how a battery works, roughly? 3 components -- two dissimilar metals and a conductive liquid. The positive metal dissolves and plates itself on the negative side(or reacts with the liquid), and an electric current flows between the two metals if joined with a wire.
One can also FORCE the reaction to happen by driving power through it. The positive side will dissolve and plate itself on the negative(or react with the liquid) as per above. If you've heard of electroplating, that's how it works.
Electrochemical machining is another use of the process. By carefully choosing the shape of the negative metal, one can electrochemically 'drill' into the positive metal. Choose the right fluid and it won't plate itself on the negative side, but just become a suspended powder; pump the fluid around to get the powder out of the way.
My first attempt at this was bill-nye-style kitchen science. Two AA batteries, a penny, and a piece of iron in saltwater with plasticine to hold stuff in place.. I screwed up, making the penny the positive side. The water rapidly filled with foamy yellow crap as the penny dissolved and ate into the zinc inside.
My second attempt reversed the two and used a copper wire instead of a penny. It slowly dug a divot into the metal. Partial success!
My third attempt was the second on a larger scale, using an insulated copper wire so as to drill a small hole instead of a wide divot. Generated hydrogen gas(this is normal) blew the varnish off the wire(good way to do it, maybe) ruining the narrow hole and digging a wide divot again.
My fourth attempt was a half-assed attempt at pumping water though the center of a gear and out the underside, and using that as the neative electrode to dig an inside gear in metal. Since it was half-assed, stuff kept moving around, I didn't have enough water for stuff to settle out, my electrolyte wasn't very good, and the pump was a retarded modified mister sprayer.
Which brings me to my fifth attempt.
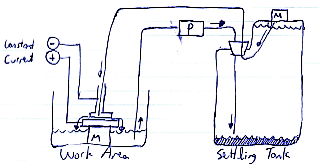
Labels for left image:
- Ancient 200W pre-ATX power supply
- Even ancienter adjustable constant-current supply, 600ma max
- Milling Machine mechanism
- Automotive windshield washer fluid pump
- 9.5L of distilled water dissolved with as much pickling salt as physically possible. That's three kilograms of salt for ten liters of water, so don't skimp when you buy.
- Rubber stopper -- may be illegal in some regions that outlaw glassware. Because only meth heads use glassware, you know. It has no legitimate uses 9.9
- Fish bubbler tubing
- The hard-to-make bit -- gear with a pipe through the hub to force water out under it. i.e. the 'cutting tool'
- A freaking plastic tub
- Cathode(-), goes to the cutting tool
- Anode(+), goes to the workpiece
- Hard-drive magnet to hold the windshield wiper motor in place
- Magnet to hold the inlet hose in place inside the plastic tank
- Duct-tape banding to hold the plastic tank in shape when it bulges disturbingly, threatening to explode and spray electrically-conductive saltwater on the perilously nearby electric outlets and power supplies(don't do what I do!)
For a bonus 500 points, find Waldo in this pic.
In the settling tank diagram, note how the input tube is way up in the top. This is so that if any bubbles get pumped in somehow, they get pushed right back out by pressure from the pump. I keep the top tube held there with a paperclip on the end and a magnet on the outside of the bottle. I like magnets. Instant fasteners without the holes or glue.
Windshield washer pumps have most impressive pressure. Even running it undervolted(5V instead of the normal 12), it bulged the settling tank in a weird and worrying way. I had to drop it to 3V before the volume of water leaving the tank balanced the volume entering. This was convenient though, since windshield washer pumps are not designed for continuous operation; even at 5V it would slowly overheat, but several hours on 3V only got it slightly warm.
Anyway, here's a bunch of images of the thing:
- Gear-shaped divot after an hour of operation
- Settling tank with mostly clear water
- Mechanism in action; the white stuff in the water is iron hydroxide, the white stuff coming up around the gear is hydrogen gas. Ventilate well!
- After 5 hours, the central hole comes free!
- The settling tank is really grungy now though it still delivered fairly clear water to the tool. I've shaken it up to dislodge the stuff sticking to the top, we'll see how well it settles over a week or three.
Conclusions: Electrochemical machining is not good for jagged shapes, it tends to round them out. It might do better for much larger gears, perhaps.


![[Use Any Browser]](/bvgraphics/enhanced.gif)